AD is a growing public health challenge, with the number of affected Americans projected to reach 8.5 million by 2030 and 13.8 million by 2060.1 Accessible and accurate tools for early detection and longitudinal studies have the potential to advance basic research and improve disease management.
Traditional AD detection requires cerebrospinal fluid samples or expensive imaging technologies, limiting access. Blood-based biomarkers like plasma phosphorylated tau 217 (p-Tau217)2,3 are an affordable alternative, offering comparable accuracy to the gold-standard methods with minimally invasive sample collection.4,5 Additionally, blood-based testing may be applied to screen broader populations before proceeding to traditional tests. Recently, the FDA approved a blood-based test for AD diagnosis, underscoring the clinical relevance of blood biomarkers.6
Advances in blood-based biomarker detection technology
Because relevant AD biomarkers are at low abundance, high analytical sensitivity is required to accurately quantify the desired proteins above background. New methods with high workflow flexibility and low barrier to entry facilitate expanded adoption.
Ultrasensitive biomarker detection
Ultrasensitive assays require a strong signal to background ratio. Several methods, including Proximity Extension Assay (PEA)7 and Nucleic acid Linked Immuno-Sandwich Assay (NULISA)8 achieve excellent sensitivity in quantifying low abundant biomarkers.
Taudia has developed the SPLASHTM (Solid Phase Ligation Assay with Single waSH) assay, which is capable of ultrasensitive protein detection without specialized instrumentation or complex workflows. Similarly to established proximity methods, the SPLASHTM platform combines dual-antibody specificity with nucleic acid-based signal amplification. No specialized instrumentation is required—standard qPCR instruments are employed to amplify the protein signals. The SPLASHTM workflow is simple yet sensitive, achieving high signal to background with a single wash step and capable of quantifying p-Tau217 at sub-picogram/mL levels, Figure 1.
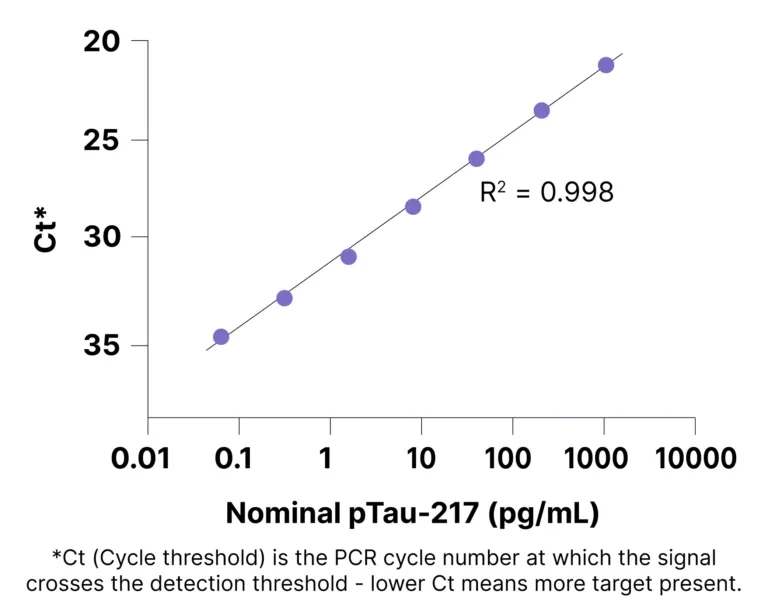
Flexible workflows for diverse research needs
AD biomarker research requires adaptable tools that accommodate varying study designs and scales. Researchers often perform large-scale screening on discovery platforms to identify candidate samples, then employ targeted assays for in-depth biomarker quantification. Taudia’s SPLASH™ assay addresses both these needs with two cost-effective workflow options.
Option 1: The benchtop workflow
The benchtop workflow (Figure 2) provides a low barrier to entry with a straightforward protocol similar to traditional immunochemistry methods like ELISA. The only requirements are standard laboratory equipment and a qPCR instrument. Results are obtained in less than four hours, with less than 30 minutes of hands-on-time. This straightforward workflow is particularly suited for laboratories seeking to implement ultrasensitive protein detection without significant infrastructure investment.
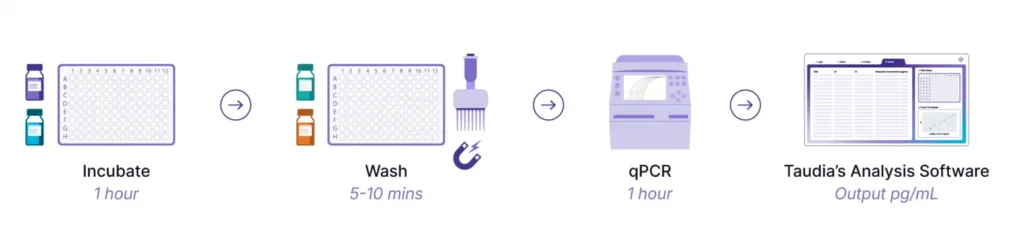
Option 2: The automated workflow
For applications demanding lower hands-on-time, researchers can seamlessly transition to Taudia’s automated workflow powered by the NeuXplore™ automated solution (Figure 3). This scalability ensures that the same assay technology can support research programs as they evolve from exploratory studies to large-scale cohort analyses, maintaining consistency in biomarker measurements across different phases of investigation.

A new era for AD research
High-sensitivity, easy-to-use blood-based biomarker detection assays empower researchers to expand AD studies and unravel the underlying biology. Blood-based biomarker detection is changing how AD research is performed—enabling fast turnaround of results, supporting earlier disease detection, and providing the opportunity to improve the future of public health.
Learn how Taudia’s ultrasensitive and flexible blood biomarker detection platform can accelerate your research.
References
- Rajan, K. B. et al. Population estimate of people with clinical Alzheimer’s disease and mild cognitive impairment in the United States (2020–2060). Alzheimer’s and Dementia 17, 1966–1975 (2021).
- Barthélemy, N. R., Horie, K., Sato, C. & Bateman, R. J. Blood plasma phosphorylated-tau isoforms track CNS change in Alzheimer’s disease. Journal of Experimental Medicine 217, (2020).
- Hansson, O., Blennow, K., Zetterberg, H. & Dage, J. Blood biomarkers for Alzheimer’s disease in clinical practice and trials. Nat. Aging 3, 506–519 (2023).
- Jack, C. R. et al. Revised criteria for diagnosis and staging of Alzheimer’s disease: Alzheimer’s Association Workgroup. Alzheimer’s and Dementia 20, 5143–5169 (2024).
- Ashton, N. J. et al. Diagnostic Accuracy of a Plasma Phosphorylated Tau 217 Immunoassay for Alzheimer Disease Pathology. JAMA Neurol. 81, 255–263 (2024).
- FDA Clears First Blood Test Used in Diagnosing Alzheimer’s Disease: New Test Provides Less Invasive Option, Reduces Reliance on PET Scans and Increases Diagnosis Accessibility. FDA News Release (2025).
- Lundberg, M., Eriksson, A., Tran, B., Assarsson, E. & Fredriksson, S. Homogeneous antibody-based proximity extension assays provide sensitive and specific detection of low-abundant proteins in human blood. Nucleic Acids Res. 39, (2011).
- Feng, W. et al. NULISA: a proteomic liquid biopsy platform with attomolar sensitivity and high multiplexing. Nat. Commun. 14, (2023).